Are these overlooked brain cells the key to treating trauma and anxiety?
A growing body of research suggests astrocytes, star-shaped brain cells once dismissed as support players, may lead the way to more precise and effective psychiatric drugs.
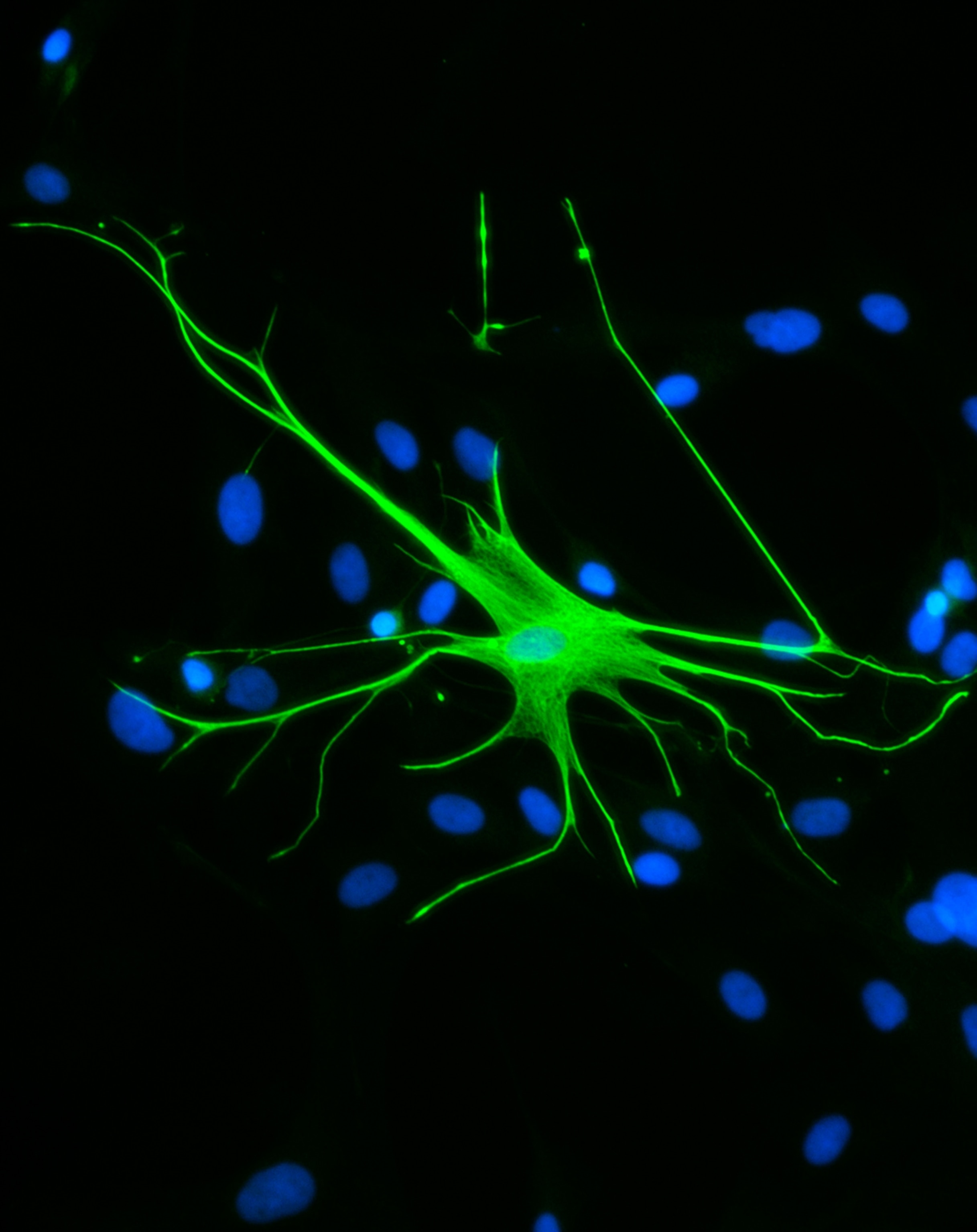
For decades, scientists have built psychiatric drugs around neurons—the brain cells that send electrical and chemical signals. But even as that understanding has deepened, treatments for mental health conditions remain frustratingly limited for the nearly one in seven people worldwide living with mental health disorders.
“The neurology and psychiatry fields have really not had a fantastic track record of treating various serious psychiatric and neurodegenerative conditions,” Anna Orr, a neuroscientist at Weill Cornell Medicine, says, “even though there’s a pretty sophisticated understanding of neuronal function.”
Now, scientists are turning to an overlooked star-shaped brain cell, the astrocyte, to develop more effective therapies. Once considered support cells, astrocytes are increasingly recognized for their role in shaping learning, memory, and behavior. Early findings suggest they could offer new, more precise targets for treating conditions like anxiety, depression, and post-traumatic stress disorder (PTSD).
How astrocytes affect brain activity and behavior
Astrocytes wrap around synapses, the junctions where neurons meet to exchange information, influencing how signals are sent and received. Although they account for as many as half of the cells in some brain regions, until recently, they were more difficult to study than neurons.
“We've had the tools for a long time to measure neural activity,” says Lindsay Halliday, a University of Arizona behavioral neuroscientist. Only in the past two decades have advances in molecular biology and genetics made it possible to track what astrocytes are doing in real time.
That shift is beginning to reveal how deeply these cells shape behavior. A recent study co-authored by Halliday showed that astrocytes in the amygdala, the brain’s emotion-processing region, lit up when mice formed and remembered fear memories. Adjusting that activity up or down led to a corresponding change in other regions of the brain, affecting how the mice reacted to fear.
“Every time we discover a new signaling pathway, we realize that the molecules involved are all genetically linked to diseases,” says Cagla Eroglu, a neuroscientist and professor at Duke University.
Astrocytes bear remarkably specific protein signatures that could make them ideal targets for new drugs. Benjamin Deneen, a neuroscientist at Baylor College of Medicine, found that a protein called NFIA remains active in astrocytes within the hippocampus, the memory region of the brain, but nowhere else. His work showed that knocking out NFIA disrupts memory and behavior in mice. “If you break an astrocyte, you impair the synapse, you alter the circuit, and behavior is eventually impacted,” says Deneen.
(Can you grow new brain cells? The debate could reveal the secret of superagers.)
Researchers are also beginning to explore whether astrocytes help explain differences in mental health between men and women. Conditions such as PTSD, anxiety, and depression are more common in women than in men. Orr identified proteins on the surface of astrocytes that behave differently in male and female brains. Activating one protein in male mice boosted memory in male mice but impaired it in females.
Understanding the sex-specific mechanisms of astrocytes opens the door to having “greater specificity,” says Orr. Once we know which pathways and which cells are driving a disease, we can intervene with treatments. However, translating findings from animal models to human treatments remains a major hurdle, and it’s still unclear how these approaches will perform in people.
Targeting the stars
Some neuroscientists believe astrocytes could lead to new classes of psychiatric medications. Severe trauma disrupts the memory and fear centers of the brain, leading people to relive vivid flashbacks of past traumatic events. Targeting the astrocytes that regulate these circuits could help reduce the impact of these memories.
(The neuroscientist working on ‘zapping’ away unwanted memories.)
Some researchers are already exploring that possibility by repurposing existing drugs. One of these drugs, KDS2010 (Tisolagiline), is in Phase 2 trials for obesity and Alzheimer’s, and may soon be tested for PTSD after promising results in mouse models of the disease. The drug works by blocking the monoamine oxidase B enzyme in astrocytes, reducing the levels of neurotransmitter GABA, which may restore healthy brain activity and reduce intrusive memories and anxiety.
Another strategy, Deneen says, involves looking for variants of genes that encode crucial astrocyte proteins that are linked to anxiety, depression, or PTSD. Researchers can test whether these variants change behavior in animals and then design drugs that tweak the same astrocyte pathways in humans.
One reason astrocytes are particularly appealing is their specificity. Many existing psychiatric drugs act broadly, affecting multiple cell types in and beyond the brain—contributing to side effects like fatigue, weight gain, and nausea. Because the proteins that regulate astrocyte activity tend to be more cell- and region-specific—and may even differ between sexes—Deneen and Orr believe that drugs that target astrocytes might have fewer side effects.
Still, much of this work remains in early stages, and translating findings from animal models to humans will take time. For now, scientists see astrocytes not as a replacement for neurons, but as part of a more complete picture of how the brain regulates memory, emotion, and behavior.